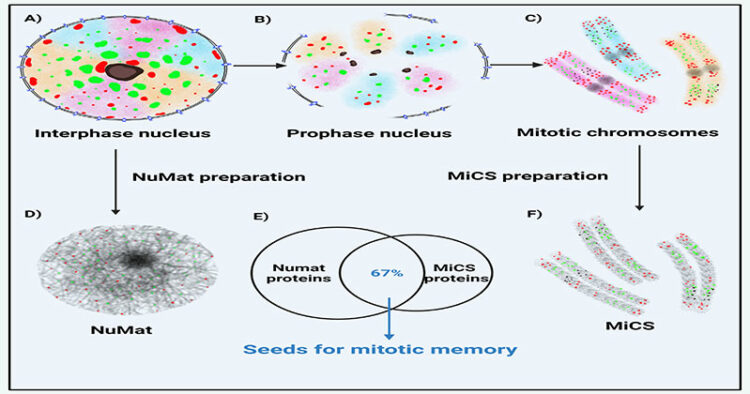
Researchers from the Centre for Cellular and Molecular Biology (CSIR-CCMB) have identified the proteins that make up the interphase nuclear scaffold called the Nuclear Matrix (NuMat) and the Mitotic Chromosome Scaffold (MiCS).
Like humans, the life of all complex organisms originates from a single cell called the zygote. During development, the cells grow in size and divide to give rise to daughter cells through mitosis. When a cell divides, the entire genetic material of the cell is replicated to make two copies and equally distributed to the daughter cells. During mitosis, the nuts and bolts of the cell and the nucleus are dismantled; after mitosis, they are automatically reassembled. While undergoing division, the cell must remember the mother cell’s organisation to reorganise the daughter cells in the same fashion.
After a particular stage of development, the fate of these cells is predetermined. A set of specific cells give rise to specific function cells in a particular organ. If and when the cells divide, their functionality must be replicated in the daughter cells. However, the mitotic memory essential for such precise functioning has yet to be discovered.
“What makes the two daughter cells identical to the mother cell is not only their physical organisation and function but also the organisation of their genetic material in the control room of the cell, the nucleus. Our lab, along with several other labs in the world, has been trying to understand the architecture of the nucleus and what could be the structural basis of mitotic memory,” said the researchers while talking to India Science Wire.
NuMat is supposed to be the underlying biomolecular scaffold that provides structural organisation and functional compartmentalisation of the nucleus. However, during cell division, the nuclear envelope and matrix essential for the structural and architectural integrity of the interphase nucleus are disassembled, and chromosome territories are condensed into mitotic chromosomes, only to be reassembled and re-established in the daughter nucleus.
“We have identified the proteins that make up the interphase nuclear scaffold called the NuMat and the MiCS. Comparing the proteins that make up the NuMat and MiCS, we found that more than two-thirds of the proteins are the same. This means, during the process of condensing the genetic material for cell division, the proteins of the nuclear matrix are packaged and passed on to the daughter cells in the form of a mitotic chromosome, which now serves as a form of mitotic memory for the precise re-establishment of mitotic memory,” researchers point out.
The researchers compared the interphase nucleus’s qualitative and quantitative protein compositions and the mitotic chromosomes from fruit fly cell lines using techniques such as Liquid Chromatography with tandem mass spectrometry (LC-MS/MS). The data analysis using bioinformatic approaches to identify the proteins in the NuMat and MiCS revealed a two-thirds overlap between them.
“Based on several studies from our lab over the years, we propose a new hypothesis to address the molecular and structural basis of mitotic memory. Our hypothesis states that the proteins that make the nuclear matrix of the interphase nucleus, during cell division are disassembled and packaged into the mitotic chromosomes and these serve as mitotic memory to guide the re-establishment of nuclear architecture in daughter cells,” researchers explain.
The study provides several new leads for a candidate-based approach to understand how the nuclear architecture dismantled during mitosis is reorganised to reproduce a cell that is structurally and functionally similar to the mother cell.
The researchers also speculate that the constituents of mitotic memory are critical for normal functioning and that their dysfunction may lead to misregulation of the cells, causing abnormalities such as cancers and developmental diseases or disorders.
The research team comprised Mamilla Soujanya, Ashish Bihani, Nikhil Hajirnis, Rashmi U Pathak, and Rakesh K Mishra. The study has been published in Springer Nature.
(India Science Wire)



















Comments